Stem Cells
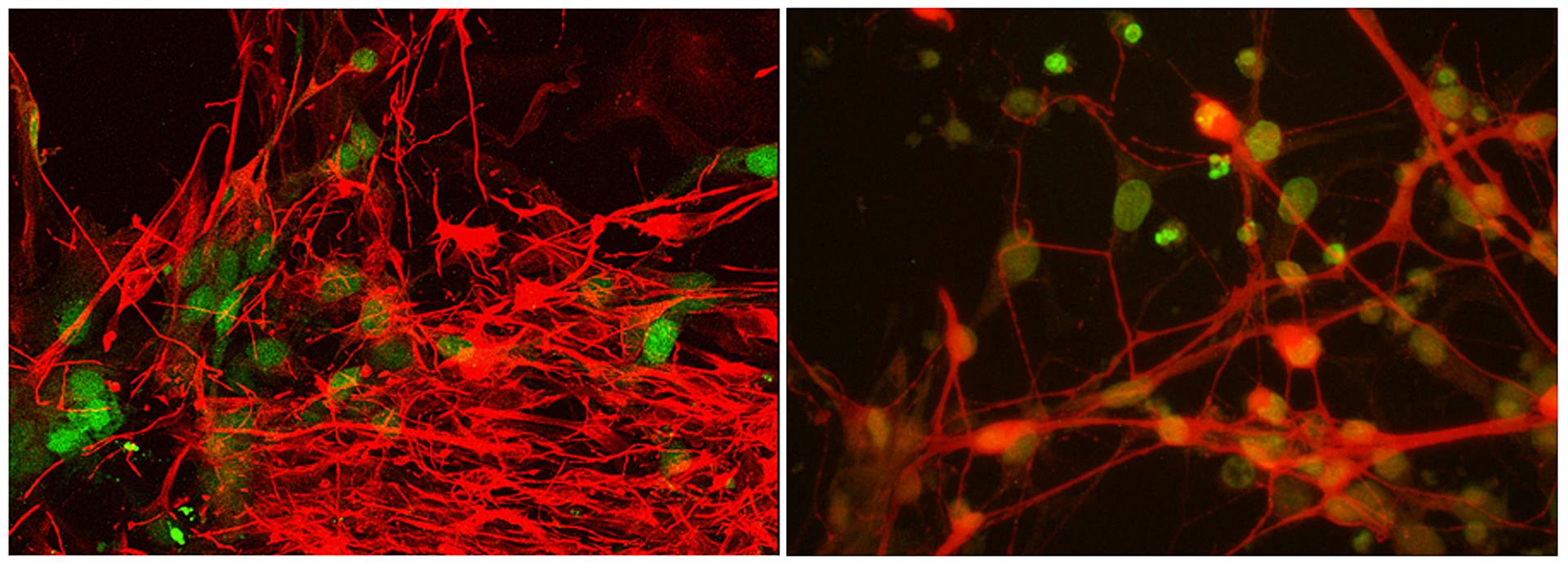
Cell-based systems in which researchers analyze the biology of a genetic variation provide tools for drug screening, and in some cases, for the creation of replacement cells and organs. In many instances, these cell-based systems will be created using patient-derived stem cells. Protocols now exist for the creation and genetic manipulation of such cells, representing the basis for individualized therapeutics of incredible sophistication and efficacy.
Columbia faculty are currently testing these concepts in areas of neurodegenerative, bone, eye, blood and cardiovascular disease, diabetes, and obesity. Very recent technical advances now permit the direct manipulation of DNA sequences in cells (DNA editing), so that mutations can be corrected, and genetic variations of interest may be introduced. Combined with this molecular technique, stem cells enable a reciprocal discovery process in which genetic analysis points to a disease mechanism, stem cells can be created from the patient, and the mutation “corrected.” These cells can be differentiated into cell types relevant to the disease to assess the validity of inferences reached on the basis of the genetic analysis. Further study of these cells can identify genes that modify the severity of the disease caused by the mutant gene. And they may lead to the identification of new drug targets.
To realize this vision, faculty clinicians and research scientists must collaborate to conduct the relevant genetic analyses and downstream stem cell-based vetting of these genes for disease mechanism and molecular interventions. To that end, the Stem Cell Core Lab, a nexus of iPS derivation, genome engineering, and directed cell differentiation of human stem cells into neuronal and non-neuronal tissues, has been created.